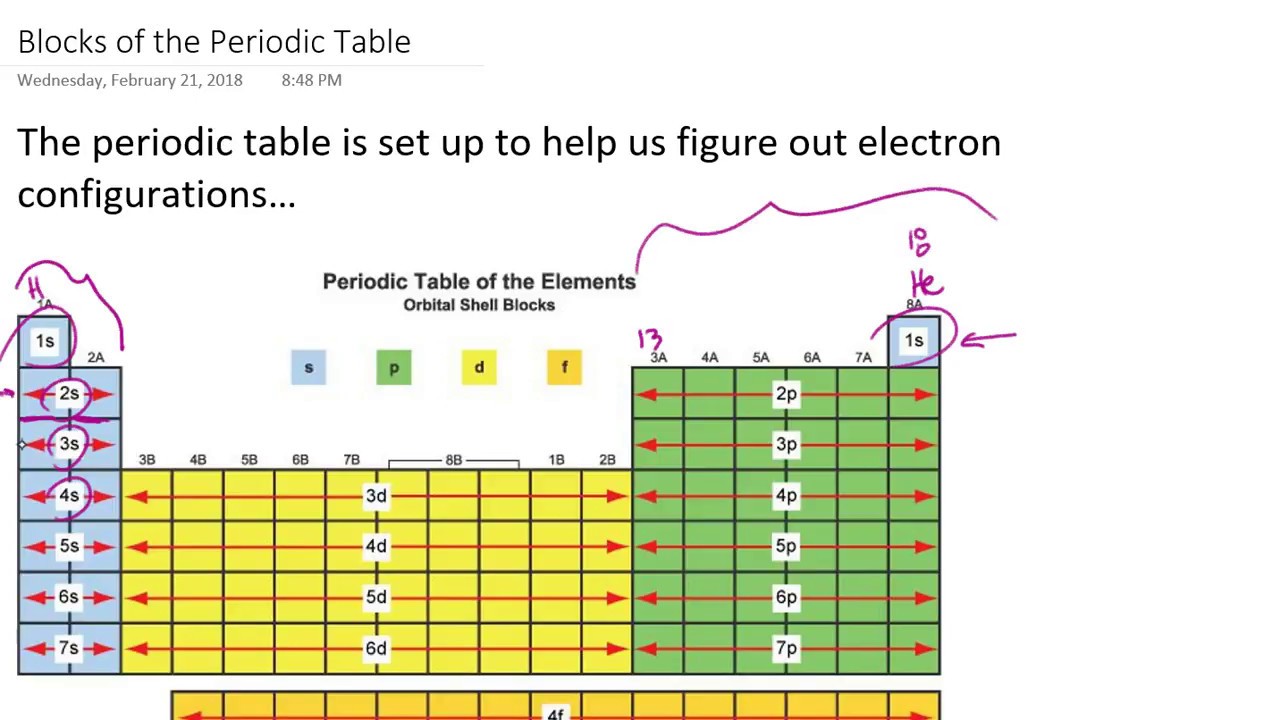
Elements in the same Group have the same number of valence shell electrons.(valence shell electrons, the number of electrons in the highest energy level, shown in red) The electrons in the valence shell (highest energy level) are given in red.Ĭan you see a pattern, or trend, in the electronic configuration of the atoms in each group and in each period? The electronic configuration in shell notation is given for an atom of each of the elements. The table below for the main group elements is set out just like the Periodic Table of the elements.
#Electron configuration periodic table free#
No ads = no money for us = no free stuff for you! Trends in the Number of Valence Electrons This article provides you with an electronic configuration chart for all these elements. Each element has a unique atomic structure that is influenced by its electronic configuration, which is the distribution of electrons across different orbitals of an atom. There are 118 elements in the periodic table. ⚛ Going down a Group of the Periodic Table from top to bottom, the energy of the valence shell electrons increases. Electron Configuration Chart for All Elements in the Periodic Table. ⚛ The valence shell electrons of elements in the same Period of the Periodic Table occupy the same energy level (shell). The truth is that atomic weights have changed as a function of time. Atomic weights found within a periodic table one might think are constant. ⚛ Going across a period of the Periodic Table from left to right the number of valence electrons increases. Such was the case when IUPAC recently reviewed elements 113, 115, 117 and 118, and decided to give them official names and symbols (goodbye, ununseptium and hello, tennessine). ⚛ Elements in the same Group of the Periodic Table have the same number of valence shell electrons (electrons in the highest energy level). According to electronic configuration, the periodic table elements are. Writing Electron Configurations Filling of Atomic. For example, the electron configuration of sodium is 1s 2 2s 2 2p 6 3s 1. Trends in number of valence electrons (electrons in the highest energy level): The electron configuration formula of elements must be connected with the periodic table. Electron configurations of atoms follow a standard notation in which all electron-containing atomic subshells (with the number of electrons they hold written in superscript) are placed in a sequence.If you consider the electronic configuration of an atom of each element in the Periodic Table you will see a number of patterns which are referred to as periodic trends, or just trends.